|
Through the end of the ANDEXXA continuous infusion. Substantial decrease in anti-FXa activity corresponding to the ANDEXXA bolus. Compared to baseline, there was a rapid and Volunteer studies and the ANNEXA-4 study in bleeding patients. 
The time course of anti-FXa activity following ANDEXXA administration was consistent among the healthy Re-elevation or Incomplete Reversal of Anti-FXa Activity Prothrombin complex concentrates, recombinant factor VIIa, or whole blood products within seven days prior Safety of ANDEXXA also has not been evaluated in patients who received The safety of ANDEXXA has not been evaluated in patients who experienced thromboembolic events orĭisseminated intravascular coagulation within two weeks prior to the life-threatening bleeding event 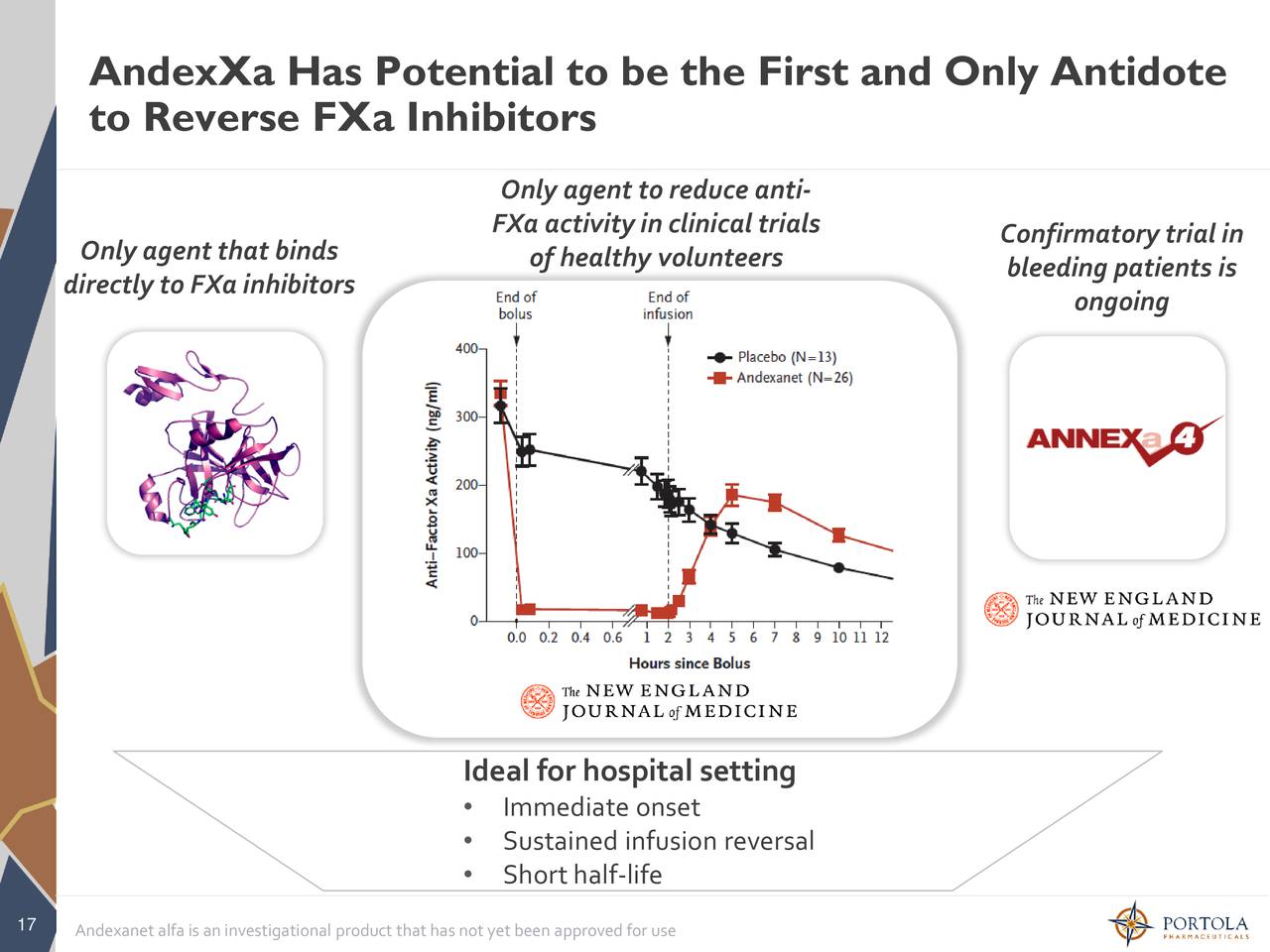
Medically appropriate following treatment with ANDEXXA. To reduce thromboembolic risk, resume anticoagulant therapy as soon as Monitor patients treated with ANDEXXA for signs and symptoms of arterial and venous thromboembolic events, Of these 223, 18 subjects (8%) had a thrombotic Of the 352 subjects who received ANDEXXA, 223 received at least one anticoagulation dose A total of 63 (18%) experienced 88 thromboembolic or Subjects who experienced a thrombotic event, the median time to first event was 7Įxperienced the event within the first three days. 
The thromboembolic and ischemic risks were assessed in 352 bleeding subjects who received ANDEXXA. Select Important Safety Information WARNINGS AND PRECAUTIONS
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
